 |
 |
- Search
| Neonatal Med > Volume 30(2); 2023 > Article |
|
Abstract
Purpose
Methods
Results
ARTICLE INFORMATION
Ethical statement
The study protocol was reviewed and approved by the Institutional Review Board (IRB) of Seoul National University Hospital (IRB No. 2206-135-1335). The requirement of informed consent requirement was waived because of the retrospective nature of the study design.
Author contributions
Conception or design: S.G.P., H.J.Y., S.Y.L., S.H.K., S.H.S., E.K.K., H.S.K.
Acquisition, analysis, or interpretation of data: S.G.P., S.H.K., S.H.S.
Drafting the work or revising: S.G.P., H.J.Y., S.Y.L., S.H.K., S.H.S., E.K.K., H.S.K.
Final approval of the manuscript: All authors read and approved the final manuscript.
Figure 1.
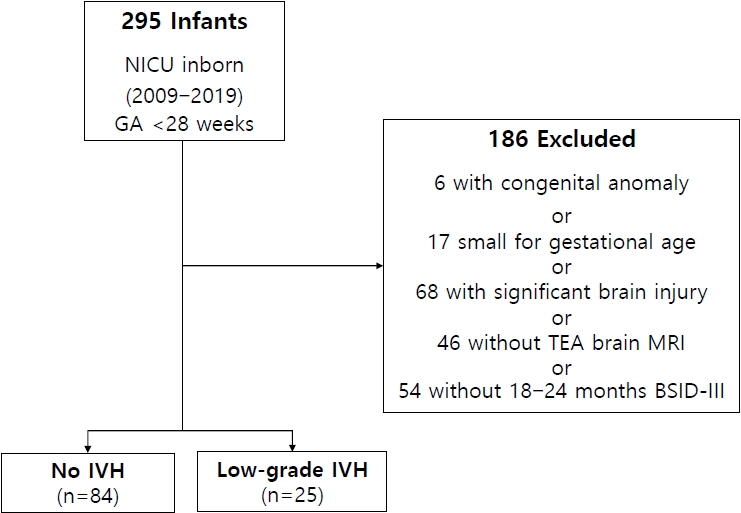
Table 1.
Table 2.
Table 3.
| Variable | aOR* | 95% CI | P-value |
|---|---|---|---|
| Ventricle (mL) | 0.985 | 0.942–1.030 | 0.508 |
| Midbrain (mL) | 1.266 | 0.807–1.986 | 0.302 |
| Pons (mL) | 1.063 | 0.809–1.398 | 0.657 |
| Medulla (mL) | 0.575 | 0.346–0.957 | 0.034 |
| Cerebellum (mL) | 0.991 | 0.960–1.023 | 0.583 |
| Thalamus (mL) | 1.054 | 0.960–1.158 | 0.268 |
| Caudate (mL) | 1.023 | 0.873–1.198 | 0.780 |
| Putamen (mL) | 1.024 | 0.848–1.237 | 0.802 |
| Pallidum (mL) | 1.051 | 0.814–1.358 | 0.700 |
| Hippocampus (mL) | 1.201 | 0.869–1.659 | 0.264 |
| Amygdala (mL) | 1.402 | 0.860–2.287 | 0.174 |
| Accumbens area (mL) | 1.263 | 0.522–3.056 | 0.601 |
| Cerebral white matter (mL) | 1.001 | 0.993–1.001 | 0.823 |
| Subcortical gray matter (mL) | 1.019 | 0.982–1.058 | 0.320 |
| Cortical gray matter (mL) | 0.996 | 0.988–1.003 | 0.222 |
| Total gray matter (mL) | 0.997 | 0.991–1.003 | 0.341 |
| Supratentorium (mL) | 0.999 | 0.996–1.003 | 0.654 |
| Total intracranial volume (mL) | 1.086 | 0.996–1.001 | 0.243 |
REFERENCES
-
METRICS

-
- 0 Crossref
- 1,549 View
- 67 Download
- Related articles in NM
-
Neurodevelopmental Outcomes of Moderate-to-Late Preterm Infants2020 November;27(4)
Neurodevelopmental Outcomes of Very Low Birth Weight Infants.2006 May;13(1)






